Mazda First Japanese Automaker To Develop Urea SCR System
HIROSHIMA, Japan�
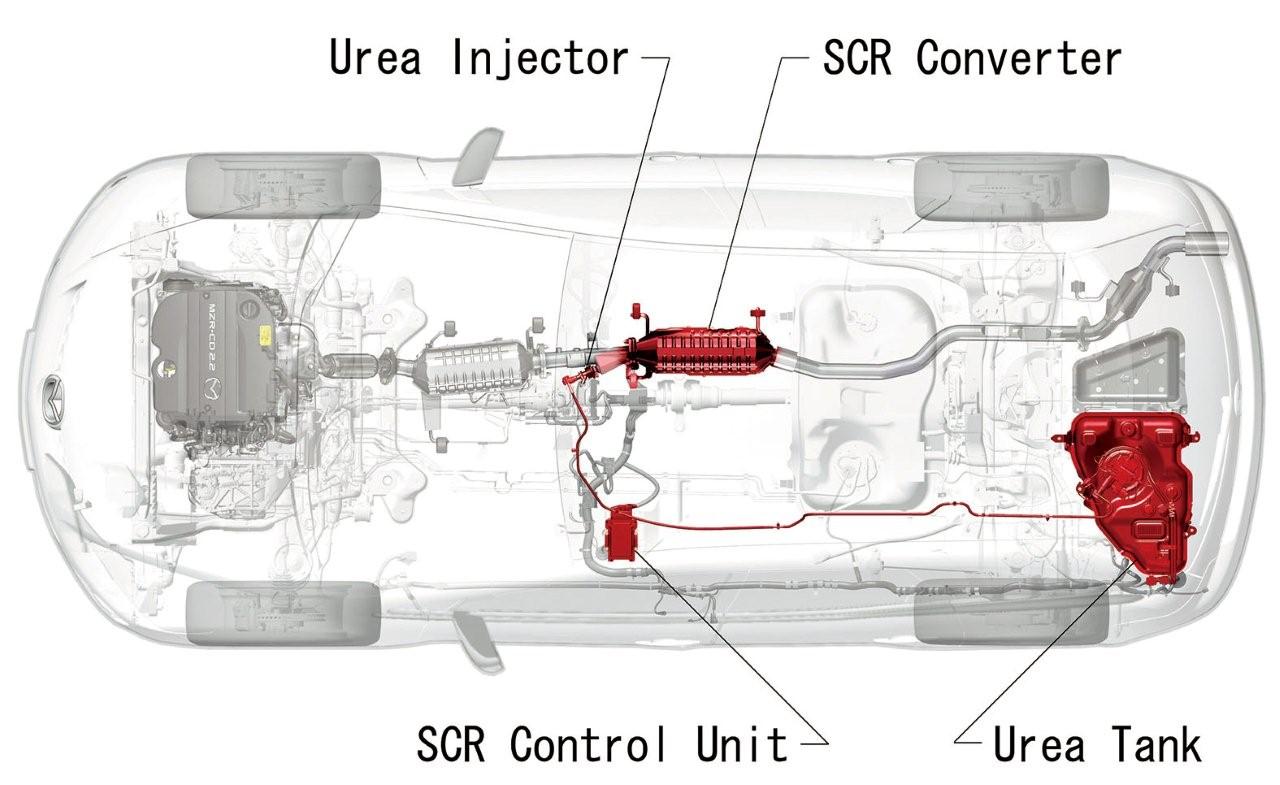
Mazda Motor Corporation�s sporty SUV, the Mazda CX-7, will be the first passenger vehicle produced by a Japanese automaker to be equipped with a urea selective catalytic reduction (SCR) system. Mazda�s SCR is smaller than previous systems, which is a significant improvement that enables it to be installed in passenger cars. It will be combined with Mazda�s newly developed clean, highly efficient MZR-CD 2.2L turbo diesel engine.
Planned for introduction in 2009, together with the new facelifted Mazda CX-7, Mazda�s SCR system will purify the nitrous oxides (NOx) that are generated in diesel exhaust gases. This will be a welcome addition to the Mazda CX-7�s new diesel engine, which provides better fuel economy and lower CO2 emissions than equivalent gasoline powerplants.
Urea SCR systems spray aqueous urea* directly into the exhaust gas flow where it purifies NOx through a chemical reaction. Unlike technologies that use extra fuel to burn off NOx, this method minimally affects fuel economy and power output. However, because an aqueous urea storage tank and other specialist equipment are required, space limitations in passenger vehicles have restricted the use of SCR systems to trucks and buses. This is not the case with Mazda�s SCR system.
Mazda�s MZR-CD 2.2L turbo diesel engine actually produces low volumes of NOx, which means that the SCR system needs to remove less NOx in post processing, resulting in a reduced amount of urea used. This means a smaller tank is required, allowing it to be comfortably installed in the Mazda CX-7.
As well as complying with Europe�s most stringent (Euro V) emissions regulations, the freshened CX-7 will provide plenty of Zoom-Zoom, perfectly balanced with eco-friendly performance.
* Urea is a harmless substance that is commonly used in lotions and hand creams.
Mazda Motor Corporation�s sporty SUV, the Mazda CX-7, will be the first passenger vehicle produced by a Japanese automaker to be equipped with a urea selective catalytic reduction (SCR) system. Mazda�s SCR is smaller than previous systems, which is a significant improvement that enables it to be installed in passenger cars. It will be combined with Mazda�s newly developed clean, highly efficient MZR-CD 2.2L turbo diesel engine.
Planned for introduction in 2009, together with the new facelifted Mazda CX-7, Mazda�s SCR system will purify the nitrous oxides (NOx) that are generated in diesel exhaust gases. This will be a welcome addition to the Mazda CX-7�s new diesel engine, which provides better fuel economy and lower CO2 emissions than equivalent gasoline powerplants.
Urea SCR systems spray aqueous urea* directly into the exhaust gas flow where it purifies NOx through a chemical reaction. Unlike technologies that use extra fuel to burn off NOx, this method minimally affects fuel economy and power output. However, because an aqueous urea storage tank and other specialist equipment are required, space limitations in passenger vehicles have restricted the use of SCR systems to trucks and buses. This is not the case with Mazda�s SCR system.
Mazda�s MZR-CD 2.2L turbo diesel engine actually produces low volumes of NOx, which means that the SCR system needs to remove less NOx in post processing, resulting in a reduced amount of urea used. This means a smaller tank is required, allowing it to be comfortably installed in the Mazda CX-7.
As well as complying with Europe�s most stringent (Euro V) emissions regulations, the freshened CX-7 will provide plenty of Zoom-Zoom, perfectly balanced with eco-friendly performance.
* Urea is a harmless substance that is commonly used in lotions and hand creams.

Urea is commercially produced from two raw materials, ammonia, and carbon dioxide. Large quantities of carbon dioxide are produced during the manufacture of ammonia from coal or from hydrocarbons such as natural gas and petroleum-derived raw materials. This allows direct synthesis of urea from these raw materials.
Commercial uses:
* A stabilizer in nitrocellulose explosives
* A reactant in the NOx-reducing SNCR and SCR reactions in exhaust gases from combustion, for example, from power plants and diesel engines
* A component of fertilizer and animal feed, providing a relatively cheap source of nitrogen to promote growth
* A raw material for the manufacture of plastics, to be specific, urea-formaldehyde resin
* A raw material for the manufacture of various glues (urea-formaldehyde or urea-melamine-formaldehyde); the latter is waterproof and is used for marine plywood
* An alternative to rock salt in the de-icing of roadways and runways; it does not promote metal corrosion to the extent that salt does
* An additive ingredient in cigarettes, designed to enhance flavour
* A browning agent in factory-produced pretzels
* An ingredient in some hair conditioners, facial cleansers, bath oils, and lotions
* A reactant in some ready-to-use cold compresses for first-aid use, due to the endothermic reaction it creates when mixed with water
* A cloud seeding agent, along with salts, to expedite the condensation of water in clouds, producing precipitation
* An ingredient used in the past to separate paraffins, due to the ability of urea to form clathrates (also called host-guest complexes, inclusion compounds, and adducts)
* A flame-proofing agent (commonly used in dry chemical fire extinguishers as Urea-potassium bicarbonate)
* An ingredient in many tooth whitening products
* A cream to soften the skin, especially cracked skin on the bottom of one's feet
* An ingredient in dish soap.
* Along with Ammonium Phosphate, as a Yeast Nutrient, for fermentation of sugars into ethanol.
* To make potassium cyanate
* A melt agent used in re-surfacing snowboarding halfpipes and terrain park features
* An nutrient used by plankton in ocean nourishment experiments for geoengineering purposes.
Urea reacts with alcohols to form urethanes. Urea reacts with malonic esters to make barbituric acids.
Commercial uses:
* A stabilizer in nitrocellulose explosives
* A reactant in the NOx-reducing SNCR and SCR reactions in exhaust gases from combustion, for example, from power plants and diesel engines
* A component of fertilizer and animal feed, providing a relatively cheap source of nitrogen to promote growth
* A raw material for the manufacture of plastics, to be specific, urea-formaldehyde resin
* A raw material for the manufacture of various glues (urea-formaldehyde or urea-melamine-formaldehyde); the latter is waterproof and is used for marine plywood
* An alternative to rock salt in the de-icing of roadways and runways; it does not promote metal corrosion to the extent that salt does
* An additive ingredient in cigarettes, designed to enhance flavour
* A browning agent in factory-produced pretzels
* An ingredient in some hair conditioners, facial cleansers, bath oils, and lotions
* A reactant in some ready-to-use cold compresses for first-aid use, due to the endothermic reaction it creates when mixed with water
* A cloud seeding agent, along with salts, to expedite the condensation of water in clouds, producing precipitation
* An ingredient used in the past to separate paraffins, due to the ability of urea to form clathrates (also called host-guest complexes, inclusion compounds, and adducts)
* A flame-proofing agent (commonly used in dry chemical fire extinguishers as Urea-potassium bicarbonate)
* An ingredient in many tooth whitening products
* A cream to soften the skin, especially cracked skin on the bottom of one's feet
* An ingredient in dish soap.
* Along with Ammonium Phosphate, as a Yeast Nutrient, for fermentation of sugars into ethanol.
* To make potassium cyanate
* A melt agent used in re-surfacing snowboarding halfpipes and terrain park features
* An nutrient used by plankton in ocean nourishment experiments for geoengineering purposes.
Urea reacts with alcohols to form urethanes. Urea reacts with malonic esters to make barbituric acids.
Urea is commercially produced from two raw materials, ammonia, and carbon dioxide. Large quantities of carbon dioxide are produced during the manufacture of ammonia from coal or from hydrocarbons such as natural gas and petroleum-derived raw materials. This allows direct synthesis of urea from these raw materials.
Commercial uses:
* A stabilizer in nitrocellulose explosives
* A reactant in the NOx-reducing SNCR and SCR reactions in exhaust gases from combustion, for example, from power plants and diesel engines
* A component of fertilizer and animal feed, providing a relatively cheap source of nitrogen to promote growth
* A raw material for the manufacture of plastics, to be specific, urea-formaldehyde resin
* A raw material for the manufacture of various glues (urea-formaldehyde or urea-melamine-formaldehyde); the latter is waterproof and is used for marine plywood
* An alternative to rock salt in the de-icing of roadways and runways; it does not promote metal corrosion to the extent that salt does
* An additive ingredient in cigarettes, designed to enhance flavour
* A browning agent in factory-produced pretzels
* An ingredient in some hair conditioners, facial cleansers, bath oils, and lotions
* A reactant in some ready-to-use cold compresses for first-aid use, due to the endothermic reaction it creates when mixed with water
* A cloud seeding agent, along with salts, to expedite the condensation of water in clouds, producing precipitation
* An ingredient used in the past to separate paraffins, due to the ability of urea to form clathrates (also called host-guest complexes, inclusion compounds, and adducts)
* A flame-proofing agent (commonly used in dry chemical fire extinguishers as Urea-potassium bicarbonate)
* An ingredient in many tooth whitening products
* A cream to soften the skin, especially cracked skin on the bottom of one's feet
* An ingredient in dish soap.
* Along with Ammonium Phosphate, as a Yeast Nutrient, for fermentation of sugars into ethanol.
* To make potassium cyanate
* A melt agent used in re-surfacing snowboarding halfpipes and terrain park features
* An nutrient used by plankton in ocean nourishment experiments for geoengineering purposes.
Urea reacts with alcohols to form urethanes. Urea reacts with malonic esters to make barbituric acids.
Commercial uses:
* A stabilizer in nitrocellulose explosives
* A reactant in the NOx-reducing SNCR and SCR reactions in exhaust gases from combustion, for example, from power plants and diesel engines
* A component of fertilizer and animal feed, providing a relatively cheap source of nitrogen to promote growth
* A raw material for the manufacture of plastics, to be specific, urea-formaldehyde resin
* A raw material for the manufacture of various glues (urea-formaldehyde or urea-melamine-formaldehyde); the latter is waterproof and is used for marine plywood
* An alternative to rock salt in the de-icing of roadways and runways; it does not promote metal corrosion to the extent that salt does
* An additive ingredient in cigarettes, designed to enhance flavour
* A browning agent in factory-produced pretzels
* An ingredient in some hair conditioners, facial cleansers, bath oils, and lotions
* A reactant in some ready-to-use cold compresses for first-aid use, due to the endothermic reaction it creates when mixed with water
* A cloud seeding agent, along with salts, to expedite the condensation of water in clouds, producing precipitation
* An ingredient used in the past to separate paraffins, due to the ability of urea to form clathrates (also called host-guest complexes, inclusion compounds, and adducts)
* A flame-proofing agent (commonly used in dry chemical fire extinguishers as Urea-potassium bicarbonate)
* An ingredient in many tooth whitening products
* A cream to soften the skin, especially cracked skin on the bottom of one's feet
* An ingredient in dish soap.
* Along with Ammonium Phosphate, as a Yeast Nutrient, for fermentation of sugars into ethanol.
* To make potassium cyanate
* A melt agent used in re-surfacing snowboarding halfpipes and terrain park features
* An nutrient used by plankton in ocean nourishment experiments for geoengineering purposes.
Urea reacts with alcohols to form urethanes. Urea reacts with malonic esters to make barbituric acids.
Bunch of damn hippies around here.
Thread
Thread Starter
Forum
Replies
Last Post
Nisaja
RX-8 Discussion
51
Sep 1, 2015 04:29 AM
akagc
RX-8's For Sale/Wanted
7
Aug 11, 2015 07:07 PM